

Do you want to certify your medical equipment to comply with relevant laws and regulations?
Do you want to certify your medical equipment to comply with relevant laws and regulations?
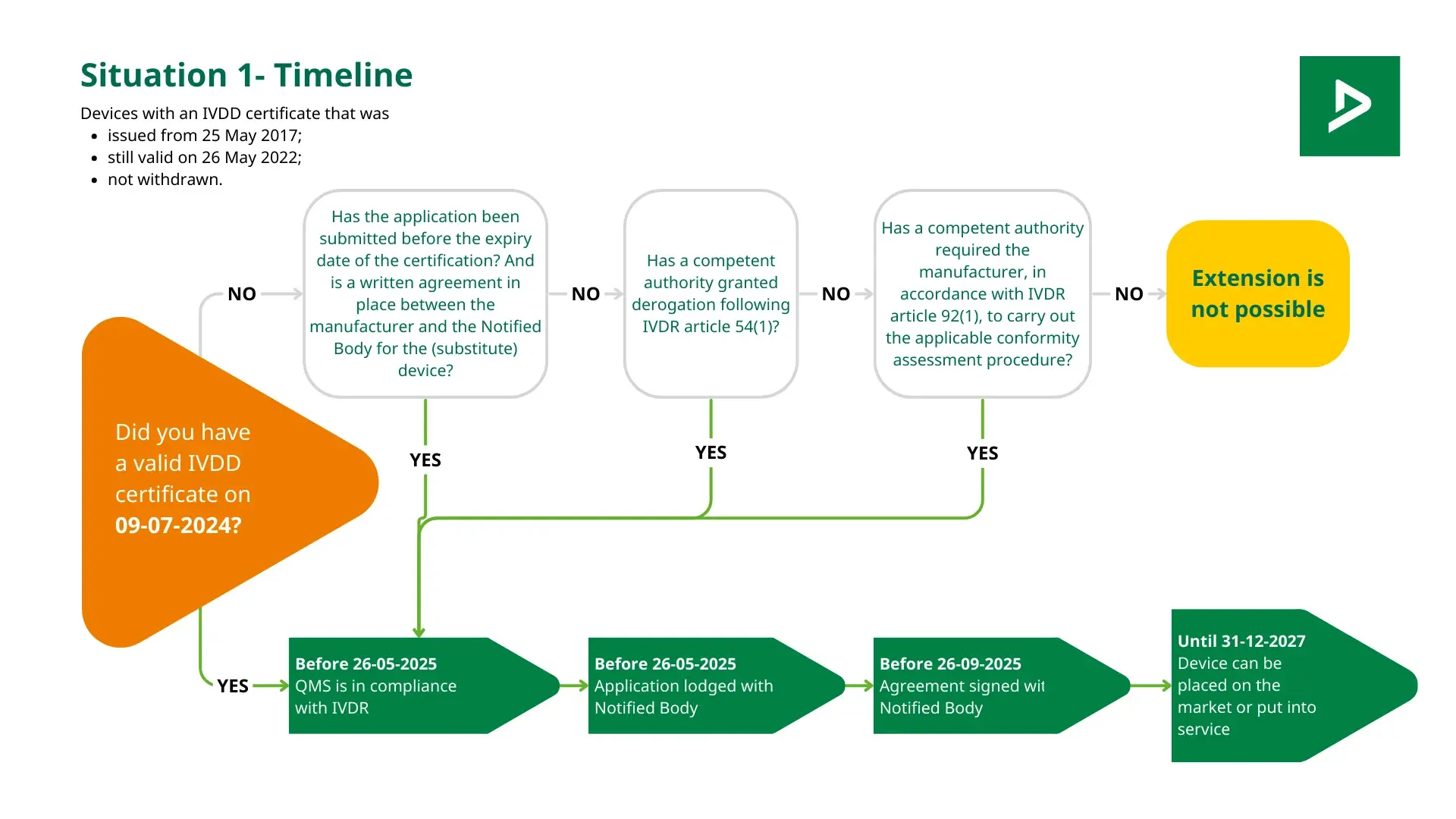
IVDR Notified Body mandatory assessment
Arrange your certification with a IVDR Notified Body
Do you want to introduce a medical in vitro diagnostic device to the European market? If so, a CE marking is required. The CE marking for in vitro diagnostic (IVD) products indicates compliance with the latest regulations, specifically the In Vitro Diagnostic Regulation (IVDR) 2017/746. Often, you will need an assessment from a notified body for this purpose. DEKRA Certification B.V. can act as a notified body to assess whether your product meets the requirements of the IVDR. Tip: initiate your CE marking application for your device under the IVDR well in advance.
Certification under IVDR: What do you need to know?

What is CE certification for medical devices?
What is a notified body?
When do you need a notified body to obtain CE IVDR certification?
What are the next steps for an IVDR assessment?
- an off-site review of the technical dossier(s);
- an on-site Quality Management System (QMS) audit.


